Table of Contents
Ever wondered how they make decaf coffee?
It is actually done through a relatively complicated scientific process known as liquid-liquid extraction (LLE).
LLE is the same process used by many industries to extract a single ingredient from a mixture. It is very challenging to master and requires precise measures to achieve optimal extraction results.
In this guide, I will help you understand the steps involved in this process. We will also discuss where exactly LLE is used in the real world and its applications.
For lab-scale understanding of the same concept, this guide shows how separatory funnel works during liquid-liquid extraction and why phase separation matters.
Ready to learn something new? Let’s get started!
What is the Liquid-Liquid Extraction Process?
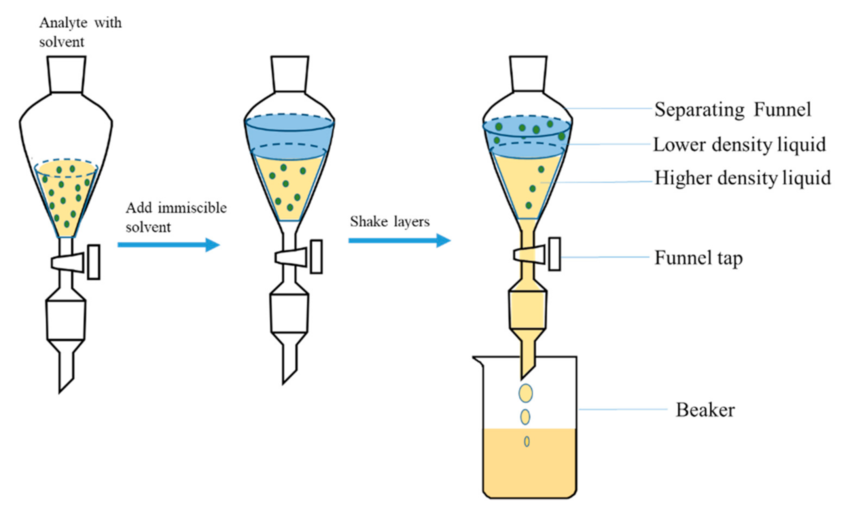
To put it simply, Liquid-Liquid Extraction (LLE) is a chemical separation method used to extract materials.
It involves adding something new to the mixture to extract the target liquid.
How does it work? The added new solvent attracts the target compound. Once the target attaches to the newly added solvent, it becomes easier to remove from the mixture.
Let’s explore the steps that show exactly how it is done in a laboratory.
Liquid-Liquid Extraction: Step-by-Step Process
For any effective separation, you need specialized equipment called a separatory funnel.
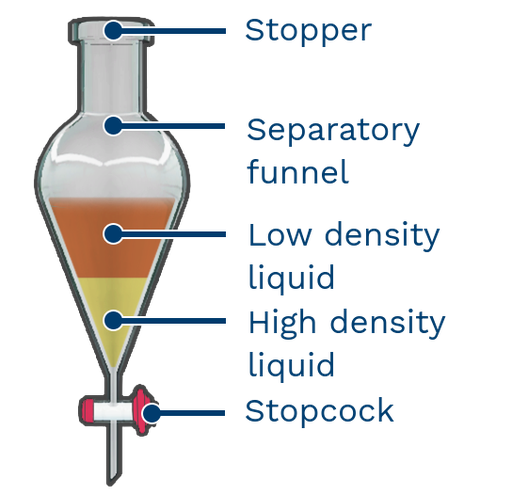
It uses the density differences between two compounds to separate them.
After stirring the funnel, the least dense liquid floats on top. Using a stopcock at the bottom, this top layer is easily separated by pouring it out from the top.
Using this funnel, we can now begin with the process of extraction using LLE:
Step 1: Choosing the Right Solvent
It is very critical to choose the perfect solvent for your extraction. This will decide the efficiency of your entire liquid-liquid extraction process.
The solvent you chose should:
- Not mix into your starting solution instead of the target compound.
- Attract the target compound to form a solution.
- Have a different density than the target compound to ensure proper purification.
- Remain easily removable after the process to obtain the target compound.
Step 2: Mixing the Solvent
This is a relatively simple step. You just need to add your chosen solvent to the separatory funnel and seal it. Then, it’s just a matter of shaking the funnel vigorously.
Note that this is not your average “making a milkshake” mixing. To separate the two original liquids, you need a lot of friction in the chamber. Only then will the target compound extract into the solvent.
To understand the science behind each step, this article on liquid liquid extraction procedure and principle explains the core mechanism in more detail.
Step 3: Let Gravity Do the Rest
It is time to let the funnel settle for a while. This helps set the solution down into two distinct layers.
Because of gravity, heavier liquids will automatically settle in the bottom layer. This layer can be either the solvent you added, mixed with your target compound, or the original liquid it was trapped in. It all depends on the type of chemical process you are currently performing.
Waiting for the solution to settle is crucial. Move the separation funnel too soon, and you may allow a cloudy layer to form between. This can further complicate your extraction.
Step 4: Separate the Layers
Once the layers have clearly formed, it’s time to separate them. This involves draining the bottom layer through the tap on the separation funnel. The topmost layer can be removed by pouring it out from the top of the funnel.
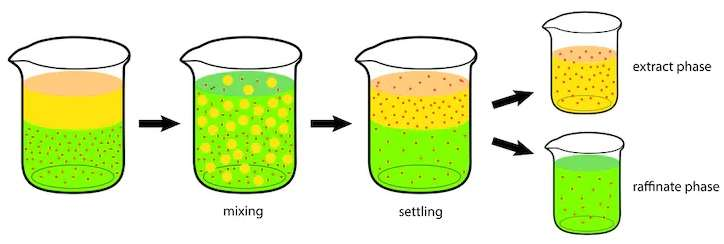
The solvent layer that you obtained from this process now includes the target compound you were after. This new layer is called the “extract”.
Step 5: Recover your target compound
Since the extract now has both your target compound and the solvent you added, it’s time to purify it. This is done by first drying the extract to remove any excess water.
Now, through evaporation or extreme heat, the solvent is removed as well. This leaves the target compound in its most purified state.
Scaling Up: From Lab to Industry
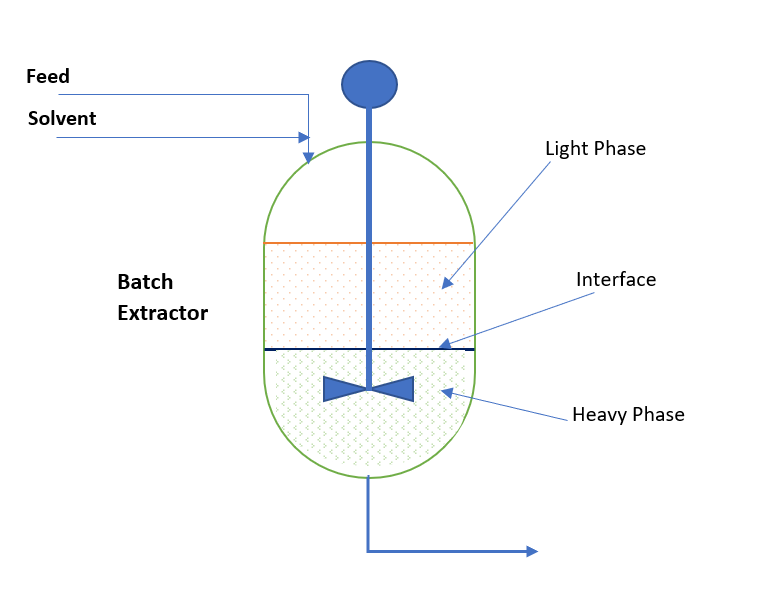
The separatory funnel used in this process is great for small-scale laboratory extractions. But doing it at large-scale factories requires specialized liquid-liquid extraction equipment.
Such equipment includes:
- Mixer-Settlers: These are industrial instruments consisting of two separate tanks. The mixed solution is kept in the first tank. It is then slowly transferred to the second tank, where it is left to separate into layers using gravity.
- Extraction Columns: In this instrument, two liquids continually flow past each other in tall towers. Since they are flowing against each other, they continue to mix and separate the target compound every time they come into contact. This process is mainly used for difficult separations that require higher energy to break the chemical bonds.
Designing a liquid-liquid extraction unit for industrial usage is not easy. It requires careful planning and an expert partner to guide you in your setup.
Real World Applications of Liquid-Liquid Extraction
LLE is not just performed in classroom laboratories. It has vital applications in the creation of everyday products. These include:
- Medicines: Used in separating antibiotics like penicillin from the broth they are fermented in.
- Coffee: The decaffeination process uses solvents or CO2 to remove the caffeine.
- Purifying Materials: Extracting contaminants from earth metals like Neodymium.
- Pollution Control: Removing impurities from industrial wastewater.
This is also one of the clearest examples of the difference between upstream and downstream processing, because extraction is mainly used during the purification stage.
Factors Affecting Liquid-Liquid Extraction Processes
There are many factors that control the efficiency of the LLE process. These include:
- pH values: Changing the pH of the target compound can make it immediately soluble in a solvent. This is a trick used by many chemists to control the reaction.
- Solvent Choice: Selecting the proper solvent that perfectly pairs with your target compound is very important for easier dissolving.
- Temperature and Time: Just like brewing tea or coffee, heat and time matter a lot in improving the quality of the transfer from one solvent to the other.
Conclusion
The liquid-liquid extraction process is one of the most widely used chemical processes. It is regularly performed on an industrial scale to address complex separation challenges.
While the principle might seem straightforward, LLE is highly complex in practice. Make one small error and the entire batch is ruined. At an industrial scale, this can lead to heavy financial losses.
To get the best advantages of liquid-liquid extraction, you need a dedicated expert like K-Jhil Scientific to get consistent results.
With over 30 years of experience in designing refining solutions for precious metals, they are genuinely the best choice. They excel at providing industry-leading equipment and engineered expertise.
This helps in making complex extraction processes safer and more reliable. With a legacy of providing gold-refining solutions to global leaders, they have already established their identity as a reliable equipment manufacturer.
If your operations involve complex recovery or extraction of precious materials, consult with the experts at K-Jhil today. They can provide you with the best path forward in implementing a more optimized and secure chemical manufacturing unit.
FAQs
1. What is the liquid-liquid extraction process?
The liquid-liquid extraction process is a separation technique in which a compound is transferred from one liquid to another. It relies on the principle that different substances dissolve more readily in different liquids, enabling purification or isolation.
2. What is a common liquid-liquid extraction example?
A classic example of liquid-liquid extraction is using ethyl acetate to isolate an organic compound, such as a flavor molecule, from an aqueous mixture. The target compound moves from the water layer into the ethyl acetate layer, where it can be easily recovered.
3. How does the solvent extraction process work?
In a solvent extraction process, a carefully chosen solvent is mixed with the original solution. The desired compound, based on its solubility, moves into the solvent. The two immiscible layers are then separated, completing the extraction.
4. What are the key factors affecting liquid-liquid extraction?
Significant factors affecting liquid-liquid extraction include the choice of solvent (its polarity and selectivity), the pH of the aqueous solution (for acids/bases), temperature, and the contact time or number of extraction stages performed.
5. What is the main equipment for liquid-liquid extraction?
The primary piece of liquid-liquid extraction equipment in a lab is a separatory funnel. For industrial scale, engineered systems like mixer-settlers, extraction columns, or centrifugal contactors form a complete liquid-liquid extraction unit.







