Table of Contents
Do you work with chemicals on a regular basis? Then you may already know how tough it can be to separate materials from a mixture.
Often, you will work with just a simple solvent with a few solids loosely suspended. But in cases of two or more liquids, the separation can get a lot tougher.
Only the right distillation system can help with this advanced separation. Choosing the best one for your needs thus becomes vital to ensure maximum efficiency.
In this guide, I will walk you through three main types of distillation. This includes:
- Simple distillation
- Fractional distillation
- Azeotropic distillation
Let’s understand and find out which suits your needs the best!
What is Simple Distillation?
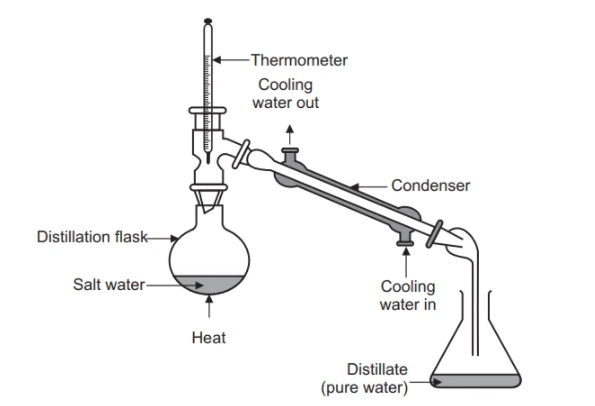
In simple distillation, we simply have to heat the mixture provided and collect the vapors. The vapors obtained on cooling will then be transferred to another container.
Simple distillation can be used when the components of the provided mixture differ in boiling points by a significant amount. Simple distillation could also be used for the extraction in chemistry of solids and liquids.
When to use this method:
- To purify water from impurities
- In order to recover non-volatile solids.
- To separate liquids with varying boiling points
- Coarse separation in which purity is not demanded.
It is important to note that, with simple distillation, we cannot extract the pure components of a given mixture, particularly when the components have close boiling points.
The components of the given mixture require a more efficient separation method to purify, and here we have the application of fractional distillation.
What is Fractional Distillation?
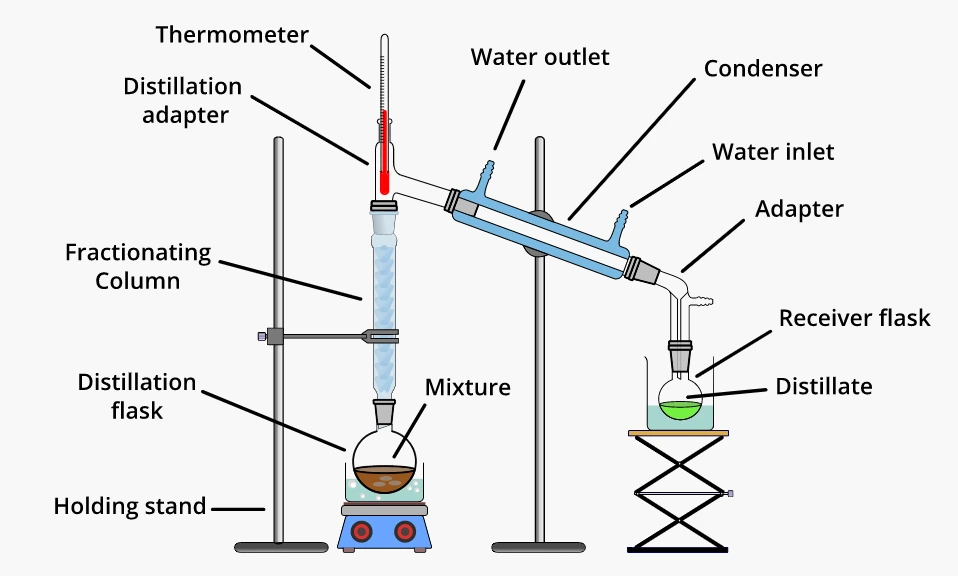
Wish to separate the components of the given mixture, but the components boil very closely together? Fractional distillation is recommended in this separation procedure.
The separation process of the components of the given mixture is performed with more accuracy and purity by fractional distillation. This is provided by the fractionating column that is situated between the flask and the condenser.
A packaging material exists in this column that helps liquid condense and evaporate several times.
In oil refineries, fractional distillation is used to separate crude oil. Chemicals in the pharmaceutical industry are also purified through this process.
When to use this method:
- Alteration of liquids of similar boiling points.
- Separating complicated mixtures and substances.
- High purity fractions
- Batch or continuous distillation
What is Azeotropic Distillation?
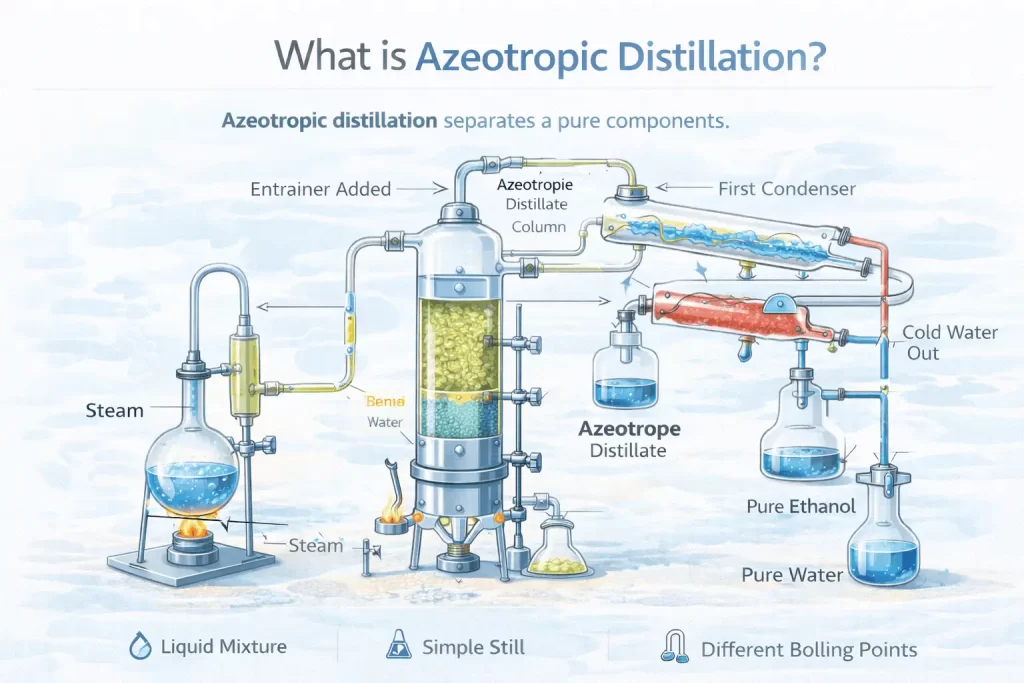
Azeotropic Distillation is a highly advanced separation technique. It helps separate components that have extremely similar boiling points.
Let’s take the ethanol-water mixture as an example. The boiling points are:
- Ethanol – 78.3°C
- Water – 100°C.
As you can see, these are very close. Moreover, these two form an azeotrope that is difficult to separate.
To separate these two materials, azeotropic distillation is required. In this method, a third substance is added to the mixture, known as the “entrainer.”
Such an entrainer is vital for breaking the chemical bonds between the two substances in the mixture.
For our ethanol-water mixture, benzene will be used to aid in its separation. It works by forming a strong bond with water. As the bond occurs, pure ethanol gets left behind.
When to use this method:
- Separating mixtures when other methods fail
- Producing 100% pure ethanol
- Separating components with very similar boiling points
- Used when conventional distillation cannot simply work
Quick Comparison: Simple vs. Fractional vs. Azeotropic
Here is how these distillation methods compare against each other:
Factor | Simple Distillation | Fractional Distillation | Azeotropic Distillation |
Boiling Point Difference | Large (>50°C) | Small to moderate (10-50°C) | Any (especially for azeotropes) |
Equipment Complexity | Low | Medium | High |
Purity Achievable | Moderate | High | Very High |
When to Use | Removing solids, rough solvent recovery | Multi-component mixtures, close-boiling liquids | Breaking azeotropes, ultra-pure products |
Industrial Example | Water purification | Crude oil refining | Absolute alcohol production |
Conclusion
Selecting the perfect industrial distillation equipment can be a tough decision. It defines the efficiency of your chemical manufacturing and requires a large investment.
To make it easier, consider these three questions before your decision:
- What is in your mixture?
For simple solids and liquids, go with simple distillation. Multiple liquids with similar boiling points require fractional distillation.
Azeotrope distillation is only needed for specialized cases.
- How much purity are you targeting?
Higher purity requires complex equipment and higher energy costs. Be very realistic about your base requirement, as many use cases do not require 100% purity.
- What is the amount of volume you need to process?
Small batches may only require simple equipment. But for high volumes, a continuous and efficient system may be required.
To ensure choosing the right fractional technique, you also need the help of a dedicated partner. A supplier that can guide you in the equipment you require for your industrial needs.
For this, I recommend partnering up with K-Jhil. They can serve as your trusted consultant and supply precise custom solutions that meet your every quality standard.
Contact the experts at K-Jhil today and receive a free consultation!
FAQs
1. What is the difference between simple distillation and fractional distillation?
The main simple distillation vs fractional distillation difference is in the equipment. Simple distillation uses just a flask and a condenser, and is good for separating liquids with large boiling-point differences (over 50°C). Fractional distillation adds a packed column between them, allowing multiple condensation cycles to separate closely boiling liquids.
2. When should I use azeotropic distillation?
Use azeotropic distillation when your mixture forms a constant-boiling azeotrope that won’t separate by normal distillation. The classic example is ethanol-water, where normal distillation stops at 95% purity. Azeotropic distillation adds an entrainer to break the azeotrope and achieve higher purity.
3. Which distillation system is best for my process?
Your distillation system choice depends on your mixture. Simple for wide-boiling or solid-liquid mixtures. Fractional for close-boiling liquids and multi-component feeds. Azeotropic for stubborn, constant-boiling mixtures that resist normal separation.
4. What’s the difference between batch and continuous distillation?
Batch vs continuous distillation comes down to operation mode. Batch processes one fill at a time. They are flexible and good for small volumes or multiple products. Continuous runs steady-state with constant feed and product removal, ideal for large volumes and consistent quality around the clock.
5. What industrial applications use fractional distillation?
Fractional distillation is everywhere in industry. Oil refineries use it to separate crude into gasoline, diesel, and kerosene. Chemical plants use it for purifying solvents and intermediates. Pharmaceutical manufacturers use it for high-purity active ingredients. It’s the workhorse of industrial distillation equipment.







