Table of Contents
In most industrial chemical processes, two reactants combine to form a third product. This output is then usually purified before being used for the final product. But what if, instead of performing these two costly steps, you could achieve both in a single reaction?
That is exactly what reactive distillation achieves. It combines chemical reactions and separations into a single unified process.
Wondering how this process works and why chemical manufacturers are rapidly adopting it?
Let’s dive deeper into reactive distillation and how it works.
What is Reactive Distillation?
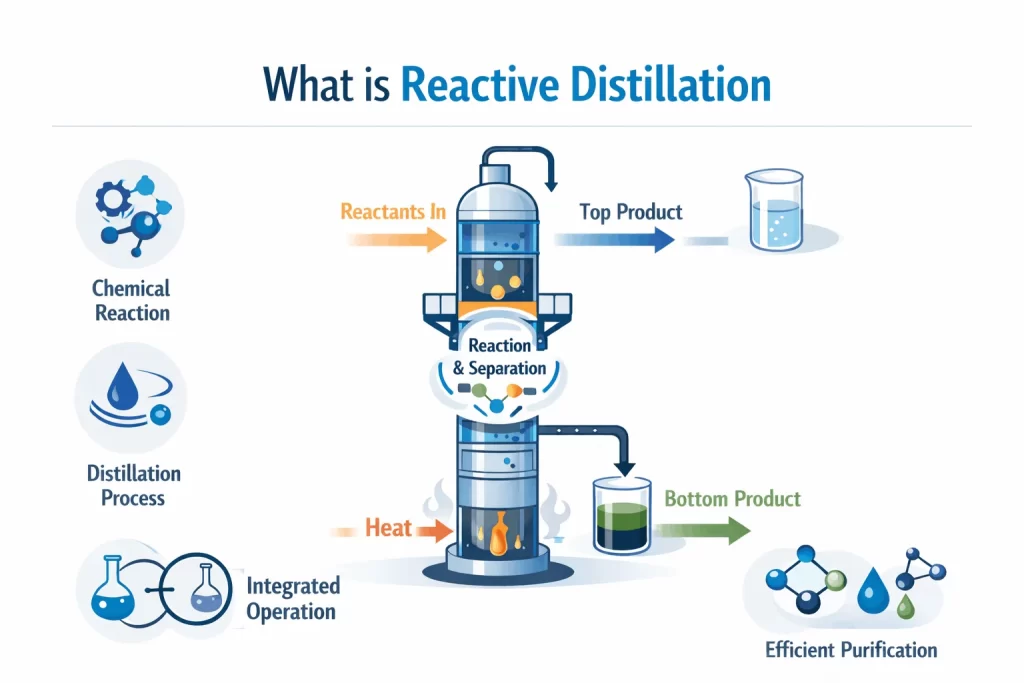
Traditionally, chemical reactions take place in a reactor. After the raw materials react with each other, the resulting material is then fed into a distillation column. This is where the products are separated from the leftovers and are purified.
To put it simply, reactive distillation enables a chemical reaction to occur inside this distillation column itself. As the products form, they are immediately separated from the mixture.
Not only does this save time and money, but it also provides a third, much more valuable advantage.
Since the products are removed, the reactants continue to react to produce more product. This is not achievable in a normal reaction, as many reactions automatically stop once enough product is formed.
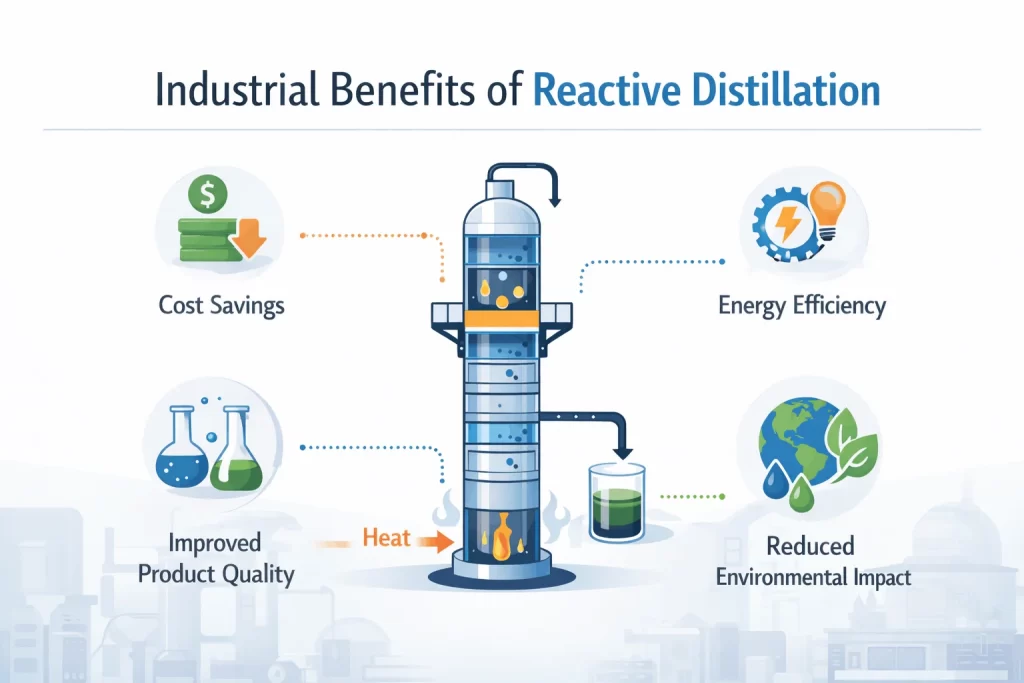
How Reactive Distillation Cuts Costs
One of the primary benefits of the reactive distillation process is the cost benefits. If you own or run a chemical plant, you can also save on the following costs:
Cost Factor | Traditional Process | Reactive Distillation | Savings |
Equipment | Separate reactor + column + reboiler + condenser | Single column unit | 20-50% lower capital cost |
Floor Space | Large footprint for multiple units | Compact design | Smaller plant footprint |
Energy Use | Heat twice (reactor + column) | Heat once | Significant energy reduction |
Recycling | Often requires recycling unreacted materials | Minimal or no recycling needed | Lower operating costs |
How Specialized Column Design Makes Reactive Distillation Possible
Wondering what makes reactive distillation possible? The secret sauce is in the special reactive distillation column design that combines two steps:
- Reaction between two or more chemicals
- Separation of products from the chamber
To achieve this, you require specialized internal components. Companies like Sulzer have developed a unique packing called Katapak™ that maximizes the reaction’s surface area.
Such packings hold a catalyst material in place while allowing vapors or liquids to pass through it. This simple design can easily scale your production from 4,000 to 50,000 tons annually.
Best Industrial Applications of Reactive Distillation
Not all processes can benefit from reactive distillation. There are specific applications where reactive distillation operates as intended.
Common reactive distillation applications include:
Application | What It Does |
Esterification | Makes esters from acids and alcohols |
Transesterification | Key process for biodiesel production |
Etherification | Produces fuel additives like MTBE |
Hydrolysis | Breaks down compounds using water |
Acetalization | Creates specialty chemicals |
How Reactive Distillation Enables Continuous Processing
One of the highlights of using this process is how it enables true continuous chemical process systems. Since the chemical reaction can continue indefinitely, you don’t need to create batch operations.
This provides benefits like:
- Gaining consistent product quality 24/7
- Lower attention requirement from operators
- Smaller equipment is required for the same output
- Easier upstream and downstream integration
Environmental Benefits of the Reactive Distillation Process
Alongside the hefty cost savings, reactive distillation also offers real environmental advantages. These include the energy savings you get by combining multiple steps.
The main benefit is the lower carbon footprint of your entire chemical production. A study found that reactive distillation can reduce CO₂ emissions by around 26.9%.
This modern technique also produces less chemical wastage. As reactions contribute entirely to creating the product, fewer byproducts remain. This eliminates the need for separate solvents or other chemical inputs.
Is Reactive Distillation Right For Your Chemical Process?
Using the right reactive distillation system can deliver consistent results with lower costs.
The process works best for chemical reactions like:
- Equilibrium-limited reactions
- Making volatile components that easily separate
- Creating products with different boiling points from the reactants
- Reactions with high energy costs and separation steps
Conclusion
Reactive distillation is one of the most successful recent improvements made in chemical processing. It allows combining two common chemical reactions in one single unit.
The evidence is clear. You get over 67% energy savings with lower costs and faster processes. These are not minor improvements, but transformative evolutions.
For efficient implementation of reactive distillation in your current manufacturing, you need the right equipment. Moreover, you will also need the expertise of a specialist in this field.
Thankfully, you have K-Jhil Scientific and its highly skilled team of experts in your corner. They bring decades of industry experience in both designing and manufacturing advanced equipment.
With ISO certification and a well-known commitment towards quality, you can always trust their advice and recommendations. Their team understands the complex needs of reactive distillation applications. This makes their implementations in your current chemical production efficient and extremely cost-effective.
Looking for a new continuous chemical processing line instead? K-Jhil’s expert team can guide you in choosing the right reactive distillation solution for your exact application. They have a proven track record of serving market leaders in chemicals, pharmaceuticals, and related industries.
Ready to explore how reactive distillation can help transform your current manufacturing efficiency? Contact K-Jhil Scientific today and get the best solution for your exact requirements.
FAQs
1. What is reactive distillation in simple terms?
Reactive distillation combines two steps into a single process. It runs a chemical reaction and separates the products simultaneously. This happens in a single column rather than on two separate machines.
2. What are the main benefits of reactive distillation?
It saves money. You need less equipment, which cuts capital costs by 20-50%. It also uses less energy, up to 67% less in some cases. This is smart chemical process optimization for any plant.
3. What industries use reactive distillation?
Many industries use it. Biodiesel makers use it for transesterification. Fuel companies use it as an additive, such as MTBE. Specialty chemical plants use it for acetalization. If your reaction hits a limit, this technology can help.
4. How is a reactive distillation column different from a normal column?
A normal column only separates mixtures. A reactive distillation column does two jobs at once. It holds a special catalyst inside while vapors and liquids flow through. The design allows reactions and separations to occur simultaneously.
5. Is reactive distillation suitable for continuous production?
Yes. Reactive distillation is perfect for continuous chemical processing. Batch reactions can take 48 hours. Continuous systems usually take much less time. You get steady quality, less labor, and more output from smaller equipment.







