Ever wondered how drinking water is extracted from seawater? What about separating the fuel we use in our vehicles from crude oil?
What's on this page:
Whether it is water or fuel, the standard process used for this extraction is distillation. But there is a difference. For extracting such components, two different types of distillation are used based on what needs to be purified. These are simple and fractional distillation.
While simple distillation is used to remove salt from seawater, the fuel we use is processed using fractional distillation. These are two industrial techniques that serve different purposes.
So what separates these two processes, and how are they practiced? Let’s go deeper to find out!
How Distillation Works?
All distillation processes work on the same simple principle: different substances boil at different temperatures.
Simple distillation involves heating the mixture to the right temperature. As the mixture contains liquids with different boiling points, this helps in extracting your target component.
It works because the liquid with the lower boiling point vaporizes. At the same time, the rest of the mixture remains in the chamber. This results in the separation of both these components.
But if both liquids have similar boiling points, extracting them by heating alone can be problematic. This is where fractional distillation comes in. It involves a complex method of repeated heating and cooling to separate the two liquids.
What are some of its other differences? Let’s explore them together.
Simple vs. Fractional Distillation
Alongside the technique, both simple and fractional distillation differ in many ways. These include:
Feature | Simple Distillation | Fractional Distillation |
Apparatus | Simple setup: distillation flask, condenser, receiver. | Includes a fractionating column between the flask and the condenser. |
Best For | Separating liquids with a significant boiling point difference ( >25°C). | Separating liquids with closer boiling points (<25°C apart). |
Purity of Distillate | Lower purity of the distillate. | Higher purity of individual liquids. |
Process Efficiency | Single distillation cycle. | Multiple, successive cycles within the chamber. |
Common Examples | Purifying seawater to get water, separating ethanol from water, etc. | Refining crude oil into gasoline, separating various components of air (oxygen, nitrogen), etc. |
The Main Difference: Fractionating Column
The most significant piece of equipment that defines fractional distillation vs distillation (simple) is the fractionating column. It is a vertical column that is packed with materials such as glass beads and metal sponges.
This fractionating column is what separates fractional distillation from simple distillation. Here is how it works:
- After heating the mixture, its vapors rise and begin cooling.
- The component that has the highest boiling point condenses first into the column.
- It drips into the column’s packing and falls back down into the mixture.
- The element with the lower boiling point remains as vapor and continues to move upward.
- This cycle keeps repeating as the vapor condenses and purifies continuously.
- Each cycle makes the mixture more purified, as the component with the lower boiling point escapes.
Simple VS Fractional Distillation (Graph Analysis)
An easy way to understand the superiority of fractional distillation is through temperature graphs. This graph plots temperature against the volume of distillate collected during the process.
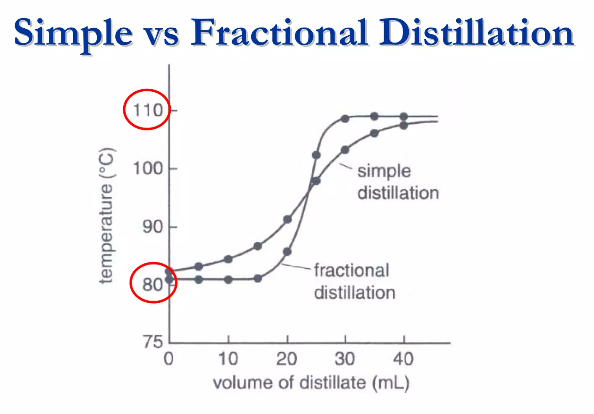
Simple Distillation Curve: It shows a gradual slope that rises with the temperature. It can only separate components if their boiling points are far apart.
Fractional Distillation Curve: It shows a curve that flattens at the boiling points of the mixture components. This is where the temperature is held steady in the chamber while a single component is collected and purified. It enables clean and efficient separation inside the mixture.
Thus, the graph clearly shows why fractional distillation is better than simple distillation.
Key Applications of Simple Distillation Vs Fractional Distillation
Now that you know how both these processes work, let’s understand how they are actually used.
Simple Distillation is used for:
- Removing salts and minerals to purify water.
- Extracting essential oils from plant material.
- Separating chemical materials that have significant boiling point differences.
Fractional Distillation is used for:
- Separating functional components, such as fuel, from crude oil.
- Producing high-purity ethanol for beverages and industrial usage.
- Purifying complex chemical compounds.
- Distilling air to produce pure nitrogen, oxygen, and argon.
Conclusion
The debate of simple distillation vs fractional distillation does not have an easy winner. While fractional distillation is more efficient, choosing between them comes down to your own industrial use case.
- Use simple distillation to separate components that have a large gap between their boiling points. It is a quick, cost-effective distillation method.
- Choose fractional distillation for handling tricky mixtures containing liquids with similar boiling points.
Mastering simple distillation vs fractional distillation is difficult in the lab. But at an industrial scale, choosing the wrong method can severely affect your profitability.
This is where you need an expert partner like K-Jhil Scientific to help increase your industrial efficiency. With a legacy of over 30 years in the refining industry, our experts can help you choose the correct distillation method.
Aside from being a global leader in gold refining, K-Jhil has proven itself as a preferred extraction partner for both small and large businesses. We can help you turn these chemical distillation principles into reliable industrial processes.
Want to get consistent results with the highest purity levels? Contact the experts at K-Jhil today!
FAQs
1. What is the key difference between simple distillation and fractional distillation?
The key difference between simple distillation and fractional distillation is the equipment. Simple distillation has a basic setup, while fractional distillation adds a special column to separate liquids with very close boiling points.
2. When is fractional distillation better than simple distillation?
Fractional distillation is better than simple distillation for separating mixtures of liquids with similar boiling points, such as those in crude oil. The extra column provides a much purer result.
3. How does a simple vs fractional distillation graph show the difference?
A simple vs. fractional distillation graph makes it clear: a simple distillation line slopes upward, indicating mixed output. A fractional graph has flat steps, proving pure substances are separated.
4. Why is fractional distillation used instead of simple distillation in industry?
Industry uses fractional distillation, not simple distillation. This is because it efficiently handles complex mixtures. The fractionating column enables multiple separations in a single step, which is crucial for products such as gasoline and jet fuel.
5. Can simple distillation separate components of crude oil effectively?
No. Separating crude oil requires fractional distillation. Simple distillation cannot effectively separate its many components, which have boiling points that are too close together.